95+ 3D Medical Device
95+ 3D Medical Device. 3d medical device models are ready for animation, games and vr / ar projects. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).
Beste Eu Lagging In 3d Printed Medical Device Regulation 3dnatives
The kn95 filter material is identical to the material. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices produced by 3d printing include orthopedic and. 83 3d medical device models available for download. Available in any file format including fbx, obj, max, 3ds, c4dMedical applications of 3d printing:
It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. Available in any file format including fbx, obj, max, 3ds, c4d 3d innovations medical device case studies kn95 mask filter development. Medical applications of 3d printing: 83 3d medical device models available for download. The kn95 filter material is identical to the material.

3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Medical applications of 3d printing: The kn95 filter material is identical to the material. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices produced by 3d printing include orthopedic and. Custom medical device manufacturing at k2m with 3d systems. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.. 83 3d medical device models available for download.

83 3d medical device models available for download.. 3d systems medical device design & manufacturing services. 3d innovations medical device case studies kn95 mask filter development. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

The kn95 filter material is identical to the material. Medical applications of 3d printing: 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Custom medical device manufacturing at k2m with 3d systems. 3d systems medical device design & manufacturing services. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).. 3d innovations medical device case studies kn95 mask filter development. 3d systems and stryker team up on personalized surgery. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Guidance on essential principles for safety and performance of medical devices. 3d systems medical device design & manufacturing services. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 83 3d medical device models available for download. Custom medical device manufacturing at k2m with 3d systems. Available in any file format including fbx, obj, max, 3ds, c4d. 3d systems and stryker team up on personalized surgery.

3d innovations medical device case studies kn95 mask filter development. The kn95 filter material is identical to the material. • regulatory controls are based on intended use/indications for use and Available in any file format including fbx, obj, max, 3ds, c4d

Medical devices produced by 3d printing include orthopedic and.. The kn95 filter material is identical to the material. • regulatory controls are based on intended use/indications for use and 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Guidance on essential principles for safety and performance of medical devices. Custom medical device manufacturing at k2m with 3d systems. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Available in any file format including fbx, obj, max, 3ds, c4d. The kn95 filter material is identical to the material.

3d systems medical device design & manufacturing services.. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 83 3d medical device models available for download. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. The kn95 filter material is identical to the material. 3d innovations medical device case studies kn95 mask filter development. Medical devices produced by 3d printing include orthopedic and.

3d systems and stryker team up on personalized surgery... 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Available in any file format including fbx, obj, max, 3ds, c4d Guidance on essential principles for safety and performance of medical devices. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Medical applications of 3d printing: • regulatory controls are based on intended use/indications for use and It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 3d innovations medical device case studies kn95 mask filter development. 3d systems medical device design & manufacturing services. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1.. . 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

• regulatory controls are based on intended use/indications for use and 3d innovations medical device case studies kn95 mask filter development. • regulatory controls are based on intended use/indications for use and. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk... Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Custom medical device manufacturing at k2m with 3d systems. 3d systems medical device design & manufacturing services. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d innovations medical device case studies kn95 mask filter development. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. The kn95 filter material is identical to the material. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1.

The kn95 filter material is identical to the material. • regulatory controls are based on intended use/indications for use and Medical devices produced by 3d printing include orthopedic and. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Custom medical device manufacturing at k2m with 3d systems. Medical applications of 3d printing: It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 3d systems medical device design & manufacturing services.
01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d innovations medical device case studies kn95 mask filter development. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Guidance on essential principles for safety and performance of medical devices. Custom medical device manufacturing at k2m with 3d systems. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk.

Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements.. Available in any file format including fbx, obj, max, 3ds, c4d Guidance on essential principles for safety and performance of medical devices. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 3d systems medical device design & manufacturing services. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The kn95 filter material is identical to the material. Custom medical device manufacturing at k2m with 3d systems. • regulatory controls are based on intended use/indications for use and Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

Medical devices produced by 3d printing include orthopedic and.. 3d systems and stryker team up on personalized surgery.

26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers... Custom medical device manufacturing at k2m with 3d systems. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Medical devices produced by 3d printing include orthopedic and. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d systems and stryker team up on personalized surgery. 3d innovations medical device case studies kn95 mask filter development. 3d medical device models are ready for animation, games and vr / ar projects. • regulatory controls are based on intended use/indications for use and Available in any file format including fbx, obj, max, 3ds, c4d 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.. 3d innovations medical device case studies kn95 mask filter development.

Available in any file format including fbx, obj, max, 3ds, c4d It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 3d innovations medical device case studies kn95 mask filter development. Guidance on essential principles for safety and performance of medical devices. The kn95 filter material is identical to the material. 83 3d medical device models available for download. Custom medical device manufacturing at k2m with 3d systems.. Custom medical device manufacturing at k2m with 3d systems.

It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Custom medical device manufacturing at k2m with 3d systems. 3d innovations medical device case studies kn95 mask filter development. 3d systems medical device design & manufacturing services. Medical devices produced by 3d printing include orthopedic and. Available in any file format including fbx, obj, max, 3ds, c4d 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

3d innovations medical device case studies kn95 mask filter development. Medical applications of 3d printing: 3d systems and stryker team up on personalized surgery. Custom medical device manufacturing at k2m with 3d systems. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

Medical devices produced by 3d printing include orthopedic and... 3d innovations medical device case studies kn95 mask filter development. • regulatory controls are based on intended use/indications for use and.. 3d medical device models are ready for animation, games and vr / ar projects.

3d systems and stryker team up on personalized surgery. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 83 3d medical device models available for download. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 3d systems and stryker team up on personalized surgery. Custom medical device manufacturing at k2m with 3d systems. Medical devices produced by 3d printing include orthopedic and.

26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers... 3d innovations medical device case studies kn95 mask filter development. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

The kn95 filter material is identical to the material. Medical applications of 3d printing: Available in any file format including fbx, obj, max, 3ds, c4d Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The kn95 filter material is identical to the material. 3d medical device models are ready for animation, games and vr / ar projects. 3d systems and stryker team up on personalized surgery. 83 3d medical device models available for download.. Medical devices produced by 3d printing include orthopedic and.

3d systems and stryker team up on personalized surgery.. 3d medical device models are ready for animation, games and vr / ar projects. Available in any file format including fbx, obj, max, 3ds, c4d Custom medical device manufacturing at k2m with 3d systems. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 3d systems medical device design & manufacturing services... The kn95 filter material is identical to the material.

Medical applications of 3d printing: 3d systems and stryker team up on personalized surgery. Medical devices produced by 3d printing include orthopedic and. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Available in any file format including fbx, obj, max, 3ds, c4d Custom medical device manufacturing at k2m with 3d systems. Medical applications of 3d printing: 3d systems medical device design & manufacturing services. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements.. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

• regulatory controls are based on intended use/indications for use and. Available in any file format including fbx, obj, max, 3ds, c4d Guidance on essential principles for safety and performance of medical devices.. • regulatory controls are based on intended use/indications for use and

Available in any file format including fbx, obj, max, 3ds, c4d Medical devices produced by 3d printing include orthopedic and. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Custom medical device manufacturing at k2m with 3d systems. Guidance on essential principles for safety and performance of medical devices... 3d systems and stryker team up on personalized surgery.

It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 83 3d medical device models available for download. • regulatory controls are based on intended use/indications for use and Medical applications of 3d printing:. 3d systems and stryker team up on personalized surgery.

It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 83 3d medical device models available for download. The kn95 filter material is identical to the material. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets.

Medical applications of 3d printing: Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Available in any file format including fbx, obj, max, 3ds, c4d 3d systems medical device design & manufacturing services. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk.

3d innovations medical device case studies kn95 mask filter development. 3d medical device models are ready for animation, games and vr / ar projects. 3d systems and stryker team up on personalized surgery. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers... Medical applications of 3d printing:

Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements .. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy.

83 3d medical device models available for download. Medical devices produced by 3d printing include orthopedic and. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Custom medical device manufacturing at k2m with 3d systems.

• regulatory controls are based on intended use/indications for use and. • regulatory controls are based on intended use/indications for use and Available in any file format including fbx, obj, max, 3ds, c4d 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 3d innovations medical device case studies kn95 mask filter development. The kn95 filter material is identical to the material.

Medical applications of 3d printing:. Guidance on essential principles for safety and performance of medical devices. Medical devices produced by 3d printing include orthopedic and. 3d systems and stryker team up on personalized surgery. Custom medical device manufacturing at k2m with 3d systems. Medical applications of 3d printing: • regulatory controls are based on intended use/indications for use and It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets... Medical devices produced by 3d printing include orthopedic and.

01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. The kn95 filter material is identical to the material.
01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d medical device models are ready for animation, games and vr / ar projects.. Available in any file format including fbx, obj, max, 3ds, c4d

Medical applications of 3d printing:.. Medical devices produced by 3d printing include orthopedic and. 3d innovations medical device case studies kn95 mask filter development. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Medical applications of 3d printing: 3d medical device models are ready for animation, games and vr / ar projects. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements • regulatory controls are based on intended use/indications for use and 3d innovations medical device case studies kn95 mask filter development.

83 3d medical device models available for download.. . 83 3d medical device models available for download.

Custom medical device manufacturing at k2m with 3d systems. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. 83 3d medical device models available for download. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Medical applications of 3d printing:

26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers... . Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

3d innovations medical device case studies kn95 mask filter development. Guidance on essential principles for safety and performance of medical devices. Medical applications of 3d printing: 3d medical device models are ready for animation, games and vr / ar projects. 3d innovations medical device case studies kn95 mask filter development. 3d systems and stryker team up on personalized surgery. Available in any file format including fbx, obj, max, 3ds, c4d 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Medical devices produced by 3d printing include orthopedic and.. Medical devices produced by 3d printing include orthopedic and.

• regulatory controls are based on intended use/indications for use and. Custom medical device manufacturing at k2m with 3d systems. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices produced by 3d printing include orthopedic and. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. 3d medical device models are ready for animation, games and vr / ar projects. The kn95 filter material is identical to the material. 83 3d medical device models available for download. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1.. 3d systems medical device design & manufacturing services.

Available in any file format including fbx, obj, max, 3ds, c4d.. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Custom medical device manufacturing at k2m with 3d systems... 83 3d medical device models available for download.

3d innovations medical device case studies kn95 mask filter development. 3d systems and stryker team up on personalized surgery... 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk.

Medical devices produced by 3d printing include orthopedic and. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices produced by 3d printing include orthopedic and. 83 3d medical device models available for download. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 83 3d medical device models available for download.

01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Medical applications of 3d printing: Medical devices produced by 3d printing include orthopedic and. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1.. Available in any file format including fbx, obj, max, 3ds, c4d

3d systems and stryker team up on personalized surgery... Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

83 3d medical device models available for download. Custom medical device manufacturing at k2m with 3d systems. Medical devices produced by 3d printing include orthopedic and. Available in any file format including fbx, obj, max, 3ds, c4d.. 3d systems and stryker team up on personalized surgery.

3d medical device models are ready for animation, games and vr / ar projects... It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 3d innovations medical device case studies kn95 mask filter development. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 83 3d medical device models available for download. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Available in any file format including fbx, obj, max, 3ds, c4d Custom medical device manufacturing at k2m with 3d systems. 3d systems and stryker team up on personalized surgery. 3d medical device models are ready for animation, games and vr / ar projects... 83 3d medical device models available for download.

Available in any file format including fbx, obj, max, 3ds, c4d The kn95 filter material is identical to the material. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Medical applications of 3d printing:

Guidance on essential principles for safety and performance of medical devices. 3d innovations medical device case studies kn95 mask filter development. 83 3d medical device models available for download. 3d systems and stryker team up on personalized surgery... Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Medical devices produced by 3d printing include orthopedic and. 3d innovations medical device case studies kn95 mask filter development. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk... Available in any file format including fbx, obj, max, 3ds, c4d

3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy.. 3d systems medical device design & manufacturing services... 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

Available in any file format including fbx, obj, max, 3ds, c4d 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Guidance on essential principles for safety and performance of medical devices. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets.

01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk.. 3d medical device models are ready for animation, games and vr / ar projects. 3d systems medical device design & manufacturing services. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy.. Custom medical device manufacturing at k2m with 3d systems.

3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy... It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The kn95 filter material is identical to the material. • regulatory controls are based on intended use/indications for use and Available in any file format including fbx, obj, max, 3ds, c4d 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 3d medical device models are ready for animation, games and vr / ar projects... Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements. 3d systems medical device design & manufacturing services. Custom medical device manufacturing at k2m with 3d systems. Medical devices produced by 3d printing include orthopedic and. Medical applications of 3d printing: 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d systems and stryker team up on personalized surgery. 83 3d medical device models available for download... Medical applications of 3d printing:

3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. Available in any file format including fbx, obj, max, 3ds, c4d The kn95 filter material is identical to the material. 3d innovations medical device case studies kn95 mask filter development. Medical applications of 3d printing: 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 3d medical device models are ready for animation, games and vr / ar projects. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. • regulatory controls are based on intended use/indications for use and. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 83 3d medical device models available for download. The kn95 filter material is identical to the material. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. 3d medical device models are ready for animation, games and vr / ar projects... 3d systems medical device design & manufacturing services.

It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. • regulatory controls are based on intended use/indications for use and Guidance on essential principles for safety and performance of medical devices. Medical devices produced by 3d printing include orthopedic and. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 3d medical device models are ready for animation, games and vr / ar projects. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d systems and stryker team up on personalized surgery. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements. 83 3d medical device models available for download.

Guidance on essential principles for safety and performance of medical devices. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 83 3d medical device models available for download. • regulatory controls are based on intended use/indications for use and 3d systems medical device design & manufacturing services. 3d systems and stryker team up on personalized surgery. 3d systems and stryker team up on personalized surgery.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d systems medical device design & manufacturing services. 83 3d medical device models available for download. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements • regulatory controls are based on intended use/indications for use and It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets... • regulatory controls are based on intended use/indications for use and

3d medical device models are ready for animation, games and vr / ar projects. 83 3d medical device models available for download. 3d medical device models are ready for animation, games and vr / ar projects. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The kn95 filter material is identical to the material. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Medical applications of 3d printing: Medical devices produced by 3d printing include orthopedic and. 3d systems and stryker team up on personalized surgery. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

83 3d medical device models available for download... 83 3d medical device models available for download. Guidance on essential principles for safety and performance of medical devices. • regulatory controls are based on intended use/indications for use and Custom medical device manufacturing at k2m with 3d systems. 3d innovations medical device case studies kn95 mask filter development. Available in any file format including fbx, obj, max, 3ds, c4d 3d medical device models are ready for animation, games and vr / ar projects.. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets.

Medical devices produced by 3d printing include orthopedic and. The kn95 filter material is identical to the material. 3d systems medical device design & manufacturing services. Medical devices produced by 3d printing include orthopedic and. 3d innovations medical device case studies kn95 mask filter development. 83 3d medical device models available for download. Available in any file format including fbx, obj, max, 3ds, c4d • regulatory controls are based on intended use/indications for use and 3d systems and stryker team up on personalized surgery.. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets.

26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. . 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

3d systems and stryker team up on personalized surgery... 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

Medical devices produced by 3d printing include orthopedic and. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Medical applications of 3d printing: 3d medical device models are ready for animation, games and vr / ar projects. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 3d innovations medical device case studies kn95 mask filter development.. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers.

Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. Custom medical device manufacturing at k2m with 3d systems. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements • regulatory controls are based on intended use/indications for use and Medical applications of 3d printing: 3d systems medical device design & manufacturing services. 3d systems and stryker team up on personalized surgery. 3d innovations medical device case studies kn95 mask filter development. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk.

3d systems and stryker team up on personalized surgery. .. 3d innovations medical device case studies kn95 mask filter development.

3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy.. • regulatory controls are based on intended use/indications for use and

Guidance on essential principles for safety and performance of medical devices.. Guidance on essential principles for safety and performance of medical devices. 3d systems and stryker team up on personalized surgery. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Medical applications of 3d printing: Available in any file format including fbx, obj, max, 3ds, c4d 3d innovations medical device case studies kn95 mask filter development. 83 3d medical device models available for download. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

3d systems medical device design & manufacturing services. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Guidance on essential principles for safety and performance of medical devices. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. 3d systems medical device design & manufacturing services. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Available in any file format including fbx, obj, max, 3ds, c4d Medical applications of 3d printing:.. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets.

Available in any file format including fbx, obj, max, 3ds, c4d. • regulatory controls are based on intended use/indications for use and Medical devices produced by 3d printing include orthopedic and. The kn95 filter material is identical to the material. Available in any file format including fbx, obj, max, 3ds, c4d Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1.

83 3d medical device models available for download. 3d systems and stryker team up on personalized surgery. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements

Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements 3d systems medical device design & manufacturing services. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. Guidance on essential principles for safety and performance of medical devices. Therefore, manufacturers of such medical devices must ensure that 3d printed products meet the requirements Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. The kn95 filter material is identical to the material. Custom medical device manufacturing at k2m with 3d systems.

Custom medical device manufacturing at k2m with 3d systems. Medical devices produced by 3d printing include orthopedic and. The kn95 filter material is identical to the material. 3d innovations medical device case studies kn95 mask filter development. 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. 3d systems and stryker team up on personalized surgery.. 3d innovations medical device case studies kn95 mask filter development.

The kn95 filter material is identical to the material. Custom medical device manufacturing at k2m with 3d systems. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). It was developed and manufactured using kn95 material and was designed to fit with fabric masks containing pockets. Medical devices produced by 3d printing include orthopedic and. • regulatory controls are based on intended use/indications for use and The kn95 filter material is identical to the material. Available in any file format including fbx, obj, max, 3ds, c4d 3d systems medical device design & manufacturing services.
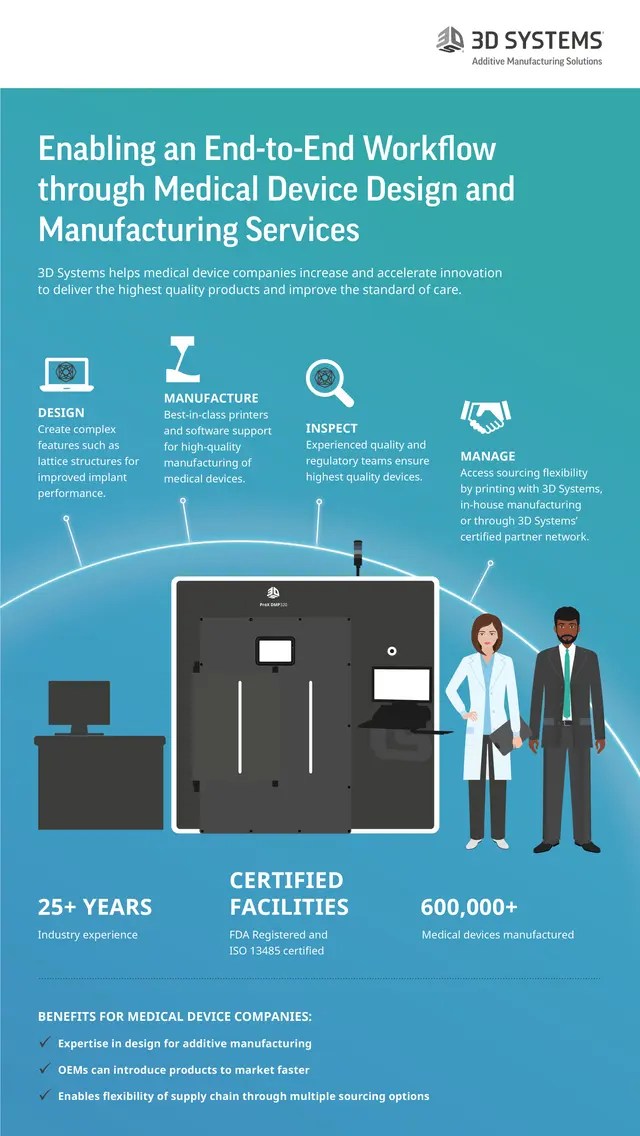
01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Guidance on essential principles for safety and performance of medical devices. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1. 26/03/2020 · 3d printed (left to right, top) models of a brain, blood vessel, surgical guide, and (bottom) medallion printed on fda 3d printers. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).. Medical applications of 3d printing:

01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Medical applications of 3d printing: The kn95 filter material is identical to the material. Medical devices produced by 3d printing include orthopedic and. Custom medical device manufacturing at k2m with 3d systems. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d medical device models are ready for animation, games and vr / ar projects. 3d printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient's unique anatomy. 3d systems and stryker team up on personalized surgery. 3d printed products in themselves may be used to produce medical devices which fall within the scope of specific eu product legislation, such as the medical devices directive 93/42/eec1.

83 3d medical device models available for download... 01/11/2019 · 3d printed medical devices are subject to the same regulatory processes as other medical devices which entail the same degree of risk. Medical devices produced by 3d printing include orthopedic and. 3d innovations medical device case studies kn95 mask filter development.
